Table of Contents
Orally dissolving remedies that contains buprenorphine are linked to intense dental issues, including complete tooth loss, the US Foodstuff and Drug Administration (Fda) warns in a security communication.
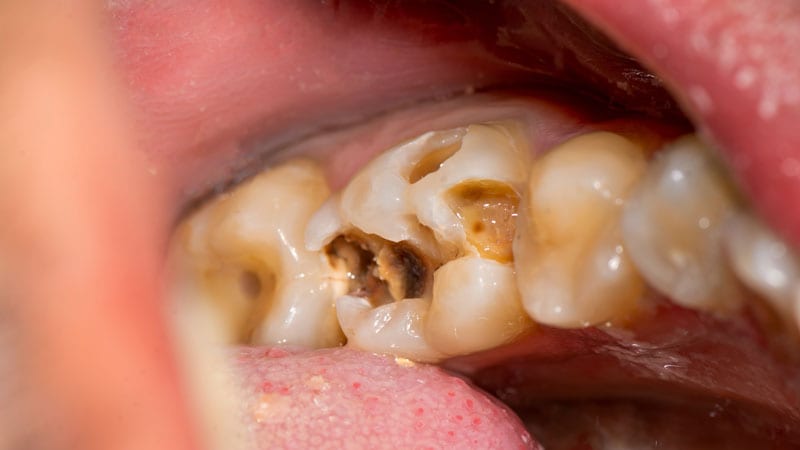
The oral aspect outcomes of these medicines, which are utilized to handle opioid use problem (OUD) and suffering, contain cavities/tooth decay, which includes rampant caries dental abscesses/an infection tooth erosion fillings slipping out and, in some circumstances, total tooth decline.
Many situations have been documented even in patients with no record of dental troubles.
The Fda is including a warning about the threat of dental difficulties to the prescribing facts and the patient medication guidebook for all buprenorphine-made up of medications dissolved in the mouth.
The Food and drug administration emphasizes, having said that, that buprenorphine remains “an crucial treatment possibility for OUD and soreness, and the benefits of these medicines evidently outweigh the threats.”
Far more Than 300 Reported Situations
Buprenorphine was permitted in 2002 as a sublingual pill, and in 2015 as a movie to be positioned inside the cheek to handle agony. Each shipping solutions have been involved with dental problems.
Considering that buprenorphine was permitted, the Food and drug administration has determined 305 cases of dental complications related with orally dissolving buprenorphine, together with 131 categorised as significant.
There may possibly be other conditions, the Food and drug administration suggests, as these signifies only circumstances claimed to the Fda or released in the professional medical literature.
The regular age of the individuals who made dental problems though getting buprenorphine is 42 yrs, but those as youthful as 18 many years previous were also affected.
Most situations transpired in individuals employing the medicines for OUD on the other hand, 28 conditions of dental troubles happened in individuals employing it to deal with agony.
In 26 conditions, individuals had no prior background of dental problems. Some dental challenges developed as soon as 2 months soon after remedy commenced the median time to prognosis was about 2 a long time right after starting treatment.
Amid all 305 instances noted, 113 associated two or more tooth.
The most popular treatment for the dental difficulties was tooth extraction/removal, which was documented in 71 circumstances. Other scenarios demanded root canals, dental medical procedures, and other strategies these kinds of as crowns and implants.
Tips
The Fda says healthcare vendors need to counsel people that critical and substantial tooth decay, tooth decline, and tooth fracture have been claimed with the use of transmucosal buprenorphine-made up of medicines and emphasize the significance of checking out their dentist to closely watch their teeth.
Sufferers need to be recommended to go on using buprenorphine remedies as prescribed and not end all of a sudden with out very first talking to their healthcare company as this could guide to significant penalties, which includes relapse, misuse or abuse of other opioids, overdose, and demise.
Patients are also becoming suggested to consider further techniques to support reduce the danger of serious dental difficulties.
Clients need to also be educated on techniques to preserve or make improvements to oral wellness when taking transmucosal buprenorphine medicines.
After the medication is fully dissolved, the individual must get a significant sip of drinking water, swish it carefully around the enamel and gums, swallow, and hold out at least 1 hour just before brushing their teeth, the Food and drug administration advises. This will let time for the mouth to steadily return to oral homeostasis and keep away from any mechanical harm that might come about owing to brushing.
The Fda also advises that people explain to their provider about any history of tooth problems, including cavities, and program a dentist pay a visit to shortly following starting the medicine.
Dental challenges connected to transmucosal buprenorphine-made up of medications need to be reported to the FDA’s MedWatch application.
For additional Medscape Psychiatry news, sign up for us on Twitter and Fb.





More Stories
Can I Get Implants with Gum Disease
Guidelines for Selecting the Ideal Pediatric Dentist for Your Child
Free smoke alarm installations and free dental screenings for children coming soon; area blood donations needed